Telehealth Kit Vendor Comparison 2026: MedConnect vs GlobalMed, TytoCare, VSee, 19Labs and More
Buying a telehealth kit in 2026 should be simple: read two or three solid vendor comparisons, cross-check the facts, request quotes from the vendors that fit your use case. Except that no serious public comparison exists for the English-speaking telehealth kit market. The pages that rank on Google for "telehealth kit" and "mobile telemedicine kit" are, almost without exception, vendor product pages. None name their competitors. None publish a criteria matrix. None list public B2B prices — only Nonagon, a consumer-grade outlier at $239.99, publishes anything. For a clinical buyer trying to choose between GlobalMed, TytoCare, VSee, 19Labs, Medpod or Visionflex, the signal-to-noise ratio is terrible.
We decided to fill that gap — under a strict rule: zero fabrication, zero editorial interpretation, only facts sourced from each vendor's public product page, with the date of consultation. If any fact below is wrong or out of date, write to us at [email protected] and we'll correct it. That's more honest than publishing nothing and leaving the market in the dark.
Upfront disclosure: we are Promotal MedConnect, so yes, we are both author and a participant in this comparison. You should read everything below through that lens. Our methodological commitment — sourced, plain-text citations, no invented claims, explicit acknowledgement of where competitors are better-fit than us — is the only thing that makes this readable rather than a sales pamphlet. Where TytoCare, GlobalMed or VSee are genuinely better positioned than us for a given buyer, we say so.
📋 Methodology and disclaimer
This comparison relies exclusively on publicly-available information on each vendor's own product pages and trust centers, consulted between April 1st and April 15th, 2026. "Not disclosed" means only that the information was not visible on the public page at the time of our consultation — it may exist in a private PDF, a signed NDA brochure, or a sales demo. We do not publish clickable outbound links to competitors; vendor names and source domains are cited as plain text. If your company is cited and you want to correct an entry, contact us.
1. The 9 vendors compared
Nine telehealth kit manufacturers or integrators serving the English-speaking market were retained for this comparison. They do not cover the entire landscape — there are dozens of device resellers, distributors and niche players — but they represent the main serious solutions visible on the first page of Google for "telehealth kit", "mobile telemedicine kit" and "portable telemedicine case" in April 2026, plus Promotal MedConnect on behalf of the European and international segments.
- Promotal MedConnect — French manufacturer (Groupe Eloi, Ernée, 97 years of medical industrial history), integrated professional kit with proprietary software platform. Deployments in France, New Caledonia, Ivory Coast, United States and Saudi Arabia (60-kit Hajj 2026 deployment in progress).
- GlobalMed — US manufacturer, one of the oldest in the segment, with dedicated product lines for disaster response, corrections, DoD/VA, offshore and school-based health. Source: globalmed.com, pages consulted April 2026.
- AMD Global Telemedicine — US manufacturer with AGNES Connect software and telemedicine bundles for rural clinics and school-based health. Source: amdtelemedicine.com, page consulted April 2026.
- TytoCare (Pro Smart Clinic) — US/Israeli vendor, the most certification-transparent player in the segment. Handheld multi-adapter architecture. Source: tytocare.com, pages consulted April 2026.
- 19Labs (GALE eClinic) — US vendor targeting rural communities, schools, workplaces, refugee and displaced-persons centers, and government health ministries. Source: 19labs.com, page consulted April 2026.
- Medpod (MobileDoc) — US vendor distributed by Henry Schein Medical, targeting Health Professional Shortage Areas, emergency departments, occupational health, schools and post-acute care. Source: medpodhealth.com, page consulted April 2026.
- VSee — US vendor with a fully documented security stack and a hardware catalog built on named third-party devices (ThinkLabs, Kardia, Interson, CloudDX, VectraCor). Real deployments on offshore oil platforms (Shell Nigeria, Alaska). Source: vsee.com/hardware, page consulted April 2026.
- Visionflex — Australian manufacturer with ProEX Mobile and the Vision Virtual Care Platform, targeting aged care, rural and remote, Indigenous health, oil and gas, merchant navy, corrections. Source: visionflex.com, page consulted April 2026.
- CureCompanion — US vendor claiming hospital-grade equipment in a ruggedised IP-67 kit with a wireless 12-lead ECG. Source: curecompanion.com/telemedicine-kit, page consulted April 2026.
Explicitly excluded from this comparison — with the reasons:
- Teladoc Health, Amwell, Caregility — not kit vendors. Teladoc Health explicitly states that its devices are "not medical devices as defined in Section 201(h) of the FD&C Act" — they're video endpoints, not clinical examination tools. Caregility ships in-room smart-room devices for hospitals, not a mobile kit. Amwell is a SaaS video platform.
- Eko, AliveCor, TytoCare Home, CliniCloud — single-device or consumer-grade products, not multi-instrument clinical kits for professional buyers.
- Nonagon Care (N9/N9+) — a consumer-grade kit retailed at $239.99. Useful context for the "kit" taxonomy, but not comparable to a professional B2B telehealth kit.
- Rijuven (Clinic in a Bag) — mentioned in the breakdown below for transparency, but their current product page is extremely thin and appears outdated. We cannot honestly matrix them against active vendors based on the April 2026 public documentation.
2. Comparison matrix — 10 clinical and compliance criteria
Every data point was verified directly on the vendor's public product page, at the date shown above. "✓ Yes" means the information is explicitly documented on the page. "— Not disclosed" means the information was not visible on the public page at the time of our consultation; it may exist elsewhere, but we will not assert it. No figures were fabricated.
| Criterion | MedConnect | GlobalMed | AMD | TytoCare Pro | 19Labs GALE | Medpod | VSee | Visionflex | CureCompanion |
|---|---|---|---|---|---|---|---|---|---|
| 12-lead ECG (standard, not optional) | ✓ Cardioline touchECG (+ Schiller, Edan) | ✓ TotalECG | ✓ Wireless ECG (brand not named) | — No ECG | ✓ QT Medical PCA-500 (via partner PR) | — Optional add-on | ✓ Kardia 12-lead | ✓ Yes (brand not named) | ✓ Wireless 12-lead |
| Digital stethoscope (brand named) | ✓ Riester Ri-Sonic | ✓ ClearSteth | ✓ Yes (brand not named) | ✓ Integrated (TytoPro handheld) | ✓ Yes (brand not named) | ✓ Yes (brand not named) | ✓ ThinkLabs | ✓ Yes (brand not named) | ✓ 100× magnification |
| Portable ultrasound | ✓ Optional | ✓ ClearProbe | ✓ Abdominal probe | — No | ✓ EchoNous POCUS (via partner PR) | ✓ Optional | ✓ Interson | ✓ Yes | — Not disclosed |
| Proprietary software platform | ✓ MedConnect | ✓ eNcounter | ✓ AGNES Connect | ✓ Tyto Insights AI | ✓ GALE platform | ✓ MobileDoc Cloud | ✓ VSee platform | ✓ ProEX / Vision Virtual Care | ✓ CureCompanion |
| AI medical scribe / automated documentation | ✓ Elara + AI SOAP scribe | — Not disclosed | — Not disclosed | ✓ Tyto Insights AI (FDA-cleared lung detection) | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed | ✓ Auto-documentation claimed |
| HIPAA BAA (stated) | ✓ Stated | ✓ Federal-grade security | ✓ Stated | ✓ Stated | — Not disclosed | — Not disclosed | ✓ Stated | — Not disclosed | — Not disclosed |
| SOC 2 Type II | — Not published | — Not disclosed | — Not disclosed | ✓ Published | — Not disclosed | — Not disclosed | ✓ Published (+ FIPS) | — Not disclosed | — Not disclosed |
| ISO 27001:2022 / ISO 13485 | ✓ ISO 27001 (June 2024) | ✓ ISO 13485 | — Not disclosed | ✓ ISO 27001 + GDPR | — Not disclosed | — Not disclosed | — Not disclosed | ✓ ISO 27001 + ISO 13485 | — Not disclosed |
| Public starting price | ✓ From ~$3,200 USD / €3,000 ex-tax | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed | — Not disclosed |
| Verified international deployment footprint | ✓ 4 continents (incl. Hajj 2026 Saudi Arabia) | ✓ "60+ countries" claimed | — Primarily US | ✓ Multi-country | ✓ Ministries, refugee centers | — Primarily US (Henry Schein) | ✓ Offshore oil (Shell Nigeria/Alaska) | — Primarily Australia | ✓ "Mobile vans globally" |
Reading the matrix: every "— Not disclosed" means only that the information was not visible on the vendor's public product page or trust center at the time of our consultation (April 2026). It may exist in a PDF brochure, a signed NDA, or a private sales deck. Our commitment is never to assert anything we cannot source to a public page.
3. Vendor-by-vendor factual breakdown
3.1 Promotal MedConnect
Positioning: integrated professional telehealth kit for European, Ministry-of-Health, humanitarian and francophone international deployments. Assembled in France in Ernée (Mayenne) by Groupe Eloi, a medical industrial group with 97 years of history.
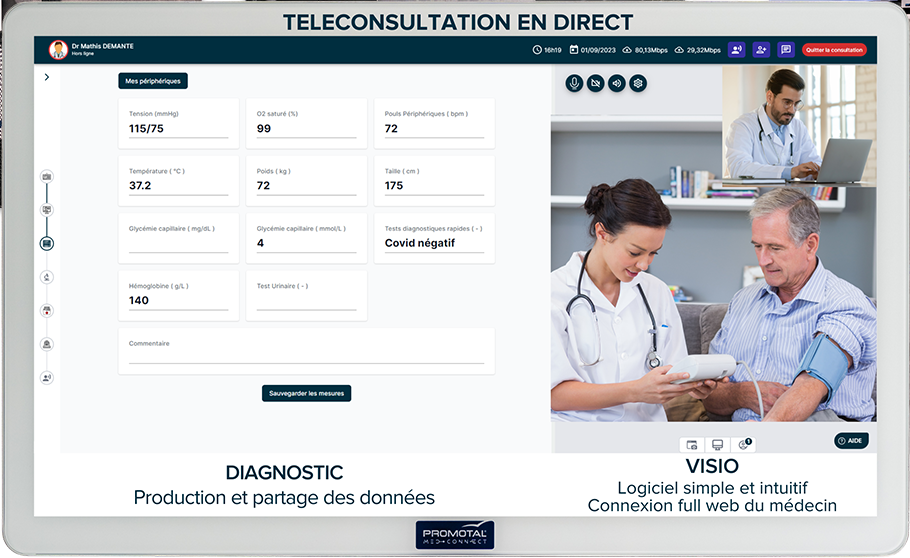
Explicitly documented on the product page: 12-lead Bluetooth ECG Cardioline touchECG (with Schiller FT1, Schiller AT2+, Edan SE-1515 as named alternatives), Riester Ri-Sonic digital stethoscope, multi-parameter bio-analyzer (glucose, cholesterol, lactate, triglycerides, uric acid), multi-lens medical camera (general practice, dermatoscope, otoscope), wireless vital signs monitor, spirometer, bladder scanner and dental camera in advanced configuration. Proprietary MedConnect software with AI medical scribe, Elara natural-language conversational agent, and access to a tele-expertise network of 110,000+ professionals. Hosted on AWS with HDS certification, ISO 27001:2022 certified since June 2024, GDPR and HIPAA compliant.
Publicly disclosed price: starting from approximately €3,000 ex-tax (~$3,200 USD) for the base configuration, scaling with added instruments.
Where we're honestly not the first-choice pick: MedConnect does not currently publish a SOC 2 Type II report — TytoCare and VSee do. We do not have US-specific FDA 510(k) clearances on every included device (we rely on CE-marking for medical-device status, which is the European equivalent, and on ISO 27001 for information security). We do not have a dedicated US DoD / federal-procurement track record — GlobalMed and AMD do. If you're a US hospital system that gates vendor selection on SOC 2 Type II, or a DoD prime contractor shopping for a NIAP-evaluated kit, evaluate those vendors first. For European deployments, Ministry-of-Health procurements and humanitarian programs, we are confident we stand up. See our MedConnect telehealth kit product page.
3.2 GlobalMed
Positioning (source: globalmed.com, pages consulted April 2026): long-established US manufacturer with dedicated product lines for disaster response, combat medicine, corrections, schools, offshore, DoD/VA. Claims 60+ countries of deployment. Product families: Transportable Exam Station (TES), Transportable Exam Backpack, Transportable Audiology Backpack, ClinicalAccess Pro Connect.
What's explicitly documented: TotalExam 3.2 Video Otoscope, TotalExam Lite, otoCam 300, Horus Series 1 and 3, MouthWatch Plus Dental Camera, ClearProbe Ultrasound, TotalVitals handheld vitals monitor, TotalECG 12-lead, Digital Spirometer, ClearSteth Stethoscope — with a claim of "55+ medical devices" integrable. Software: eNcounter (proprietary). ISO 13485 certification mentioned, FDA-clearance referenced (without 510(k) numbers on the public page), federal-grade security language, CAC/PIV reader support.
What's not disclosed on the public page as of April 2026: no SOC 2 Type II, no ISO 27001, no public pricing, no FHIR/HL7 certification level surfaced.
Best-fit use case: US DoD, VA, corrections, federal procurement, emergency response, disaster response. The strongest vendor for any buyer whose procurement gates on federal security standards and a long track record in the US defense ecosystem.
3.3 AMD Global Telemedicine
Positioning (source: amdtelemedicine.com, page consulted April 2026): US manufacturer targeting remote clinical providers and rural school-based clinics. Software-forward with AGNES Connect. Configurable bundles rather than a single SKU.
What's explicitly documented: medical cameras and scopes (otoscopy, dermatology, ophthalmology), interactive digital stethoscopes, vital signs (SpO2, BP, temperature), digital spirometer, wireless ECG, abdominal ultrasound probe, intraoral dental camera, digital colposcope. AGNES Connect cloud platform, described as encrypted. HIPAA compliance stated.
What's not disclosed on the public page as of April 2026: no SOC 2 Type II, no ISO 27001, no specific FDA 510(k) numbers, no public pricing, no named stethoscope brand, no AI scribe, no hard-case SKU (the offer is "customizable bundles"). The public instrument spec is thin relative to GlobalMed or VSee.
Best-fit use case: US rural clinics and school-based health programs looking for a flexible, customizable bundle architecture with solid US vendor presence but without the DoD footprint of GlobalMed.
3.4 TytoCare (Pro Smart Clinic)
Positioning (source: tytocare.com, pages consulted April 2026): US/Israeli vendor with the most certification-transparent stack in the segment. Architecture: a single handheld device (TytoPro) with interchangeable adapters for ear, throat, heart, lungs, skin, abdomen, temperature, plus BP, SpO2 and weight peripherals.
What's explicitly documented: Tyto Insights AI with an FDA-cleared lung Wheeze / Crackle / Rhonchi detection algorithm (cleared July 2024), EHR real-time sync, integration with Teladoc Health. Full compliance stack published publicly: HIPAA, SOC 2 Type 2, ISO 27001, GDPR, FDA-cleared medical device. This is the most complete public cert stack we observed in the entire international comparison.
What's not on the product page as of April 2026: no 12-lead ECG (the kit has no ECG at all), no ultrasound, no bio-analyzer, no spirometer. The architecture is deliberately narrow — a single examination device plus a few peripherals — not a broad multi-instrument clinical station. No public pricing.
Best-fit use case: US hospital systems, community health centers and senior care facilities where the key procurement gate is certification transparency (HIPAA BAA + SOC 2 Type II + FDA clearance), not instrument breadth. TytoCare is the benchmark any other vendor should aspire to on the cert-publication front.
3.5 19Labs (GALE eClinic)
Positioning (source: 19labs.com, page consulted April 2026): US vendor targeting rural communities, schools, workplaces, pharmacies, refugee and displaced-person centers, and government health ministries. Three product lines: GALE eClinic Point-of-Care, GALE|Schools, GALE|Toci (WhatsApp AI). This is the vendor whose target customer profile overlaps most closely with MedConnect's international Ministry-of-Health and humanitarian positioning.
What's explicitly documented: "25+ smart diagnostic devices" (page language). Via partner press releases: QT Medical PCA-500 12-lead ECG, EchoNous POCUS ultrasound. Proprietary telemedicine platform with deployment management, dashboards and SOPs. Zoom integration referenced in a customer story.
What's not on the page as of April 2026: the instrument list is vague on the vendor's own site (you have to triangulate via partner press releases to find specific brands and models). No compliance information published (HIPAA BAA, SOC 2, ISO 27001 — none surfaced on the pages we consulted). No AI scribe. No public pricing. No FDA 510(k) details on the public pages.
Best-fit use case: international government health ministry procurements, refugee and humanitarian deployments, public-health programs in emerging markets. The product catalog is right-sized for these use cases — but serious institutional buyers will need to request detailed compliance documentation that is not on the public site today.
3.6 Medpod (MobileDoc)
Positioning (source: medpodhealth.com, page consulted April 2026): US vendor distributed by Henry Schein Medical, targeting Health Professional Shortage Areas, emergency departments, occupational health, schools, urgent care and post-acute care. Positioned as "The Ultimate Portable Practice".
What's explicitly documented: vitals monitor, scale, stethoscope, general exam camera with three lenses (general, otoscope, dermatoscope). Optional add-ons include EKG, spirometry, hearing/vision screener, retinopathy, ultrasound, ABI, colposcope, dental lens and portable X-ray. Cloud platform with two-way televideo, medical-grade data streams, bidirectional EMR integration, and a 17,000+ backup physician network.
What's not on the public page as of April 2026: no HIPAA BAA claim, no SOC 2, no ISO 27001, no FDA 510(k) references, no public pricing. Most notably, ECG is optional, not standard — a serious limitation for chest-pain triage use cases. The Henry Schein distribution channel is an advantage for US procurement logistics.
Best-fit use case: US organizations already working with Henry Schein for medical supplies that want to extend their procurement relationship into telehealth hardware with minimal vendor friction. Budget-conscious US deployments where ECG is not a day-one requirement.
3.7 VSee
Positioning (source: vsee.com/hardware, page consulted April 2026): US vendor with the most transparent security disclosure in the segment, using third-party devices exclusively (rather than proprietary hardware). Real-world deployments on Shell offshore oil platforms (Nigeria, Alaska), traveling nurses, home care providers. Product lines: Telemedicine Kit, Home Care Kit, Telemedicine Cart.
What's explicitly documented: the Telemedicine Kit specifies every OEM by name — Lenovo X12 tablet, Firefly scopes, ThinkLabs digital stethoscope, Interson ultrasound (vascular/abdominal), CloudDX SpO2+BP, Kardia single-lead and 12-lead EKG, VectraCor USB spirometer. The Home Care Kit lists iHealth SpO2+BP+glucometer, AliveCor single-lead EKG, ThinkLabs One, TemporaLogix otoscope, DermLite dermatoscope — all in a ~4 lb package. Security stack published publicly: HIPAA, 256-bit AES FIPS, DTLS 1.2+, SOC 2 Type II.
What's not on the page as of April 2026: no ISO 27001, no FDA 510(k) claim on the kit itself (the kit uses FDA-cleared third-party devices — a valid but indirect compliance architecture), no AI medical scribe, no proprietary hardware, no public pricing.
Best-fit use case: offshore energy, remote industrial deployments, traveling-nurse agencies, and any buyer who wants full transparency on OEM devices inside the kit. If you want to audit every component against its own FDA clearance, VSee is the most verifiable vendor in the comparison.
3.8 Visionflex
Positioning (source: visionflex.com, page consulted April 2026): Australian manufacturer targeting aged care, rural and remote, Indigenous health, in-home care, GP, oil and gas, merchant navy, armed forces and corrections. Product lines: ProEX Mobile (kit), All-in-One Virtual Care Cart, Telehealth Mobile Kit.
What's explicitly documented: digital stethoscopes, ECG, video otoscopes, exam cameras, dental cameras, BP, SpO2, thermometer, endoscopes, ultrasound, glucose meters, video exam glasses, scales. Vision Virtual Care Platform and ProEX Clinical Software. ISO 27001 and ISO 13485 certifications mentioned, with TGA/FDA compliance referenced.
What's not on the page as of April 2026: no SOC 2 Type II, no named stethoscope or ECG brand on the public pages, no AI scribe, no public pricing. US distribution is weaker than home-market US vendors — most recent case studies are Australian or Pacific.
Best-fit use case: APAC deployments, Indigenous health programs in Australia and New Zealand, aged care facilities across the Commonwealth, and buyers who want a TGA-regulated vendor with a clear ISO 27001 stack but without a US federal-procurement track record.
3.9 CureCompanion
Positioning (source: curecompanion.com/telemedicine-kit, page consulted April 2026): US vendor marketing a "hospital-grade equipment in a lightweight kit" with an IP-67 ruggedness rating. Target: rural and community health, urgent care, schools, in-home visits, mobile vans "globally".
What's explicitly documented: digital stethoscope with 100× magnification, handheld exam camera, digital otoscope (1280×1080), Bluetooth SpO2, Bluetooth BP, wireless 12-lead ECG, ophthalmoscope / fundus camera, glucometer, Bluetooth lung monitor, JEDMED Horus Scope. CureCompanion telehealth platform with AI workflow and auto-documentation claimed. IP-67 ruggedness rating. "Clinically validated" language.
What's not on the page as of April 2026: no FDA 510(k) numbers, no HIPAA BAA statement, no SOC 2 Type II, no ISO 27001, no public pricing, no named cloud host. Device total weight (16-20 lb per public spec) is heavier than VSee's 4 lb Home Care Kit — the IP-67 rugged case is a spec trade-off.
Best-fit use case: rugged field deployments, mobile medical vans, urgent-care units where physical durability (IP-67 rating) and a wireless 12-lead ECG at an accessible price point matter more than the full cert-publication transparency of TytoCare or VSee. A direct spec-for-spec competitor to MedConnect on the instrument axis — minus the proprietary AI scribe + tele-expertise network + published ISO 27001 cert.
3.10 Rijuven (Clinic in a Bag)
Positioning (source: rijuven.com, page consulted April 2026): US vendor targeting remote communities, post-acute care, chronic disease management and senior care. Product: "Clinic in a Bag" platform + CardioSleeve (3-lead EKG + digital auscultation + Systolic Time Interval) + Rejiva EKG.
Caveat: the current public product page is extremely thin. Most instrument and feature references we found are in a 2016 press release rather than the current site. The site itself appears dated (last visible updates predate 2023). We include Rijuven here for completeness but we cannot honestly place them in the matrix above with confidence.
Recommendation: if Rijuven interests you, contact them directly to obtain a current product brochure and verify commercial status before engaging a procurement process.
4. Which vendor fits which use case?
This section does not replace a quote or a sales demo. It synthesises, from the facts documented above, the scenarios where each vendor appears best-positioned — and those where they are less so. We deliberately do not always recommend MedConnect.
- US hospital system / health system, SOC 2 Type II required in procurement: TytoCare (full cert stack, FDA-cleared AI) or VSee (HIPAA + SOC 2 Type II + FIPS) are the most procurement-ready choices. MedConnect does not currently publish a SOC 2 Type II report.
- US DoD, VA, federal procurement, corrections: GlobalMed is the default choice — 60+ countries, federal-grade security language, CAC/PIV reader support, long track record. AMD is a softer US-federal alternative. Visionflex is the APAC equivalent.
- US rural clinic, HRSA-funded, ECG required: CureCompanion (wireless 12-lead ECG, IP-67) and VSee (Kardia 12-lead, ThinkLabs stethoscope, full security stack) are strong options. Medpod is compelling if you already buy from Henry Schein and can live with ECG as an optional add-on.
- US school-based health, occupational health, urgent care: AMD, Medpod and 19Labs GALE|Schools all target this vertical explicitly. Medpod's Henry Schein distribution is a procurement advantage.
- Remote offshore (oil platforms, maritime, remote industrial): VSee has the most public reference deployments (Shell Nigeria, Alaska). GlobalMed's Transportable Exam Station is also well-positioned with its federal heritage. MedConnect has Nouvelle-Calédonie and Ivory Coast deployments in similar connectivity conditions.
- International Ministry-of-Health, humanitarian, refugee or displaced-person programs: 19Labs GALE is explicitly positioned here. MedConnect is the strongest alternative with verified deployments on four continents including the 60-kit Hajj 2026 program in Saudi Arabia and the Ivory Coast async-mode deployment (150 patients screened per day). See our international solutions.
- European hospital, primary care network, or long-term care facility: MedConnect is the most complete choice — 12-lead Cardioline touchECG, Riester Ri-Sonic stethoscope, bio-analyzer, multi-lens camera, spirometer, bladder scanner, proprietary platform, AI scribe, 110,000+ tele-expertise network, AWS HDS hosting + ISO 27001:2022. Assembled in France by a 97-year-old industrial group. The French market has its own parallel set of vendors (Tessan, HEALPHI, PARSYS, etc.) which we compare in the French-language edition of this post.
- APAC, Australia, New Zealand, aged care, Indigenous health: Visionflex is the obvious regional choice. TytoCare is the cert-first alternative if your procurement cares more about security transparency than APAC presence.
5. Where MedConnect stands in this comparison
As the author of this article, the honest positioning statement:
MedConnect is the only vendor in this comparison that simultaneously publishes — on its public product page — a 12-lead Bluetooth ECG with a named manufacturer (Cardioline touchECG with Schiller and Edan as documented alternatives), a named digital stethoscope (Riester Ri-Sonic with eMurmur AI compatibility), a multi-parameter bio-analyzer with the exhaustive list of analyses, an AI medical scribe with a conversational Elara agent, a 110,000+ tele-expertise network, an explicitly named AWS host with HDS certification, ISO 27001:2022 certification since June 2024, and a public starting price (~$3,200 USD / €3,000 ex-tax). No single competitor in the matrix above publishes all eight elements together.
MedConnect is also the only vendor with a documented Hajj 2026 deployment (60 kits in progress in Saudi Arabia), deployments in Côte d'Ivoire and Nouvelle-Calédonie in austere connectivity conditions, and more than 50,000 recorded examinations on the platform to date. For Ministry-of-Health procurements, humanitarian programs, and francophone international deployments, this body of evidence is difficult to match.
Where MedConnect is honestly not the strongest pick today:
- SOC 2 Type II: not currently published on our trust center. TytoCare and VSee are ahead of us on this specific procurement gate, which matters for many US health systems. This is on our roadmap; if a SOC 2 Type II report is a procurement blocker for you today, TytoCare or VSee are valid alternatives.
- US DoD / federal procurement: we do not have a federal-contractor track record comparable to GlobalMed. If you are a DoD prime, evaluate GlobalMed first.
- US health-system vendor relationships: we do not have a Henry Schein-level US medical-supply distribution partnership. Medpod is more convenient to procure if you already buy via Henry Schein.
- Device-level FDA 510(k): we rely on CE-marking for medical-device status on bundled instruments (the European equivalent) plus ISO 27001:2022 for information security. TytoCare publishes individual FDA 510(k) clearances on its AI algorithms, which we do not yet match.
If any of those four points are a hard procurement gate for your organization, a competitor in this comparison may serve you better. For everyone else — European deployments, Ministry of Health buyers, humanitarian and francophone international programs, and buyers who value published ISO 27001 + named AWS hosting + AI scribe + the broadest instrument catalog + public pricing — MedConnect is, in April 2026, the vendor we can document most thoroughly.
6. Next steps
Before deciding, three concrete actions:
- Read our 2026 telehealth kit buyer guide — it contains the 12-criterion checklist that lets you evaluate any vendor beyond this comparison.
- Ask every shortlisted vendor the same structured questions: (a) exact list of included instruments with brand and model, (b) name of the cloud host + data residency, (c) dated copy of their SOC 2 Type II or ISO 27001 certificate, (d) signed HIPAA BAA (if US), (e) all-inclusive 3-year total cost (hardware + software + maintenance). A vendor evasive on any of these points is a signal.
- Download the Promotal MedConnect catalog as a PDF if you want to compare our datasheet against competitor responses — no sales call involved.
7. Frequently asked questions
Why doesn't this comparison include Teladoc Health, Amwell or Caregility?
These vendors are not telehealth kit makers. Teladoc Health explicitly states that its devices "are not medical devices as defined in Section 201(h) of the FD&C Act" — they're video endpoints, not clinical examination tools. Caregility ships in-room smart devices for hospital rooms (ICU, NICU), not a mobile clinical kit. Amwell is a SaaS teleconsultation platform. None of them integrate an ECG, stethoscope, or bio-analyzer in a mobile multi-instrument case format, so they do not address the same clinical need.
Is the matrix biased in favor of MedConnect?
We are MedConnect — so the question is legitimate. Our methodological commitment: every "✓ Yes" for MedConnect can be verified on our telehealth kit product page, our software platform page and our Vanta trust center. Every "— Not disclosed" for a competitor means only that the information was not visible on their public product page in April 2026 — not that the capability does not exist. Section 5 also explicitly lists the four criteria where MedConnect is honestly not the strongest pick today, and recommends specific competitors for those cases. If a competitor believes we have missed factual information, we will correct on request at [email protected].
Why is no vendor publishing public B2B pricing?
The pattern is universal across the segment. Our research found exactly one vendor publishing anything — Nonagon at $239.99, which is a consumer product (not B2B). Every B2B telehealth kit vendor gates pricing behind "contact sales". The reasons are partly commercial (vendors adjust pricing by customer profile — federal, health system, international, humanitarian), partly operational (kit configurations are highly variable, so a single "list price" rarely maps to what a buyer actually receives). MedConnect publishes a base starting price (~$3,200 USD / €3,000 ex-tax) because we believe the information asymmetry currently hurts buyers. This is an outlier in the segment.
What about Nonagon, Eko, AliveCor, TytoCare Home?
These are either consumer-grade kits (Nonagon at $239.99) or single-device products (Eko CORE stethoscope, AliveCor KardiaMobile ECG, TytoCare Home). They're useful in their own right, but they don't fit the definition of a multi-instrument B2B clinical kit. We exclude them from the matrix to keep the comparison apples-to-apples.
How does the French telehealth kit market compare to the English-speaking one?
Very differently. The French market is dominated by a smaller set of players — Tessan, HEALPHI, PARSYS — with fewer than ten serious vendors. The English-speaking market is more fragmented (9+ vendors in this comparison alone), more certification-conscious (HIPAA BAA + SOC 2 Type II are procurement gates), and more federally oriented (GlobalMed, AMD and Visionflex explicitly target DoD / corrections / federal). If you're evaluating the French market specifically, see our French-language comparison post covering the French vendors.
Will this comparison be updated?
Yes. We revise this type of content at least twice a year and accept corrections or additions from vendors we missed. If your company is cited and you want to correct, complete or withdraw your profile, write to [email protected] — we will document the update with the new consultation date.
8. Methodology disclaimer
This comparison is an editorial / journalistic work intended for informational purposes, written by the Promotal MedConnect team in April 2026. Information about competitors comes exclusively from their public product pages and trust centers, consulted between April 1st and April 15th, 2026. "Not disclosed" signals only that the information was not visible on the public page at the time of consultation — it may exist in a PDF brochure, a signed NDA, or a private sales deck. Brand and product names remain the property of their respective owners. No external clickable link points to any competitor website, in order to respect their commercial independence and avoid ambiguous association. If you are a cited vendor and you want to correct, complete or withdraw your profile, please write to [email protected] — we will document the update with the date.
2. Comparison matrix — 10 clinical and technical criteria
Every data point was verified directly on the vendor's public product page, at the date shown above. "✓ Yes" means the information is explicitly documented on the page. "— Not mentioned" means the information was not visible on the public page at the time of our consultation; it may exist elsewhere, but we will not assert it. No figures were fabricated.
| Criterion | MedConnect | Tessan | HEALPHI | PARSYS | Medeo Kligo | HOPI Wallet | MediSpot ONE | Hocobag |
|---|---|---|---|---|---|---|---|---|
| 12-lead ECG | ✓ Cardioline touchECG (+ Schiller, Edan) | — Not mentioned | — Not mentioned | ✓ Yes (Bluetooth) | ✓ Compatible (catalog) | — Not mentioned | — Not mentioned | — Page inaccessible |
| Digital stethoscope | ✓ Riester Ri-Sonic | ✓ Yes (model not specified) | ✓ Yes (model not specified) | — Not mentioned | ✓ Compatible (catalog) | ✓ Yes | ✓ Cardio-pulmonary | — Page inaccessible |
| Multi-parameter bio-analyzer | ✓ Glucose, cholesterol, lactate, triglycerides, uric acid | — Not mentioned | — Not mentioned | ✓ Glucometer (option) | ✓ Compatible (catalog) | — Not mentioned | — Not mentioned | — Page inaccessible |
| Proprietary integrated software | ✓ MedConnect | ✓ Tessan platform | ✓ Healphi Web (+ Doctolib / Maiia / Qare compat.) | ✓ Parsys Cloud / MedCapture | ✓ Kligo (+ EHR integrations) | — Third-party compat. only | ✓ MediSpot.Center | — Page inaccessible |
| AI medical scribe (auto SOAP notes) | ✓ Yes (Elara + AI Scribe) | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Page inaccessible |
| Integrated tele-expertise network | ✓ 110,000+ professionals | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Page inaccessible |
| Named HDS-certified host | ✓ AWS (HDS-certified) | — Not mentioned | ✓ "HDS servers" (host not named) | ✓ "HDS" (host not named) | — Not mentioned | — Not mentioned | — Not mentioned | — Page inaccessible |
| ISO 27001:2022 | ✓ Certified since June 2024 | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Not mentioned | — Page inaccessible |
| Public starting price | ✓ From €3,000 ex-VAT | — Not disclosed | — "Cheapest on the market" (no figure) | — Not disclosed | — Not disclosed | — Not disclosed | ✓ Online shop | — Page inaccessible |
| Assembly in France | ✓ Ernée (Mayenne) — Groupe Eloi | — Not specified | ✓ Claimed | ✓ Claimed | — Not specified | — Not specified | — Not specified | — Page inaccessible |
Reading the matrix: every "— Not mentioned" means only that the information was not visible on the vendor's public product page at the time of our consultation (April 2026). It may exist in a PDF brochure, a private quote, or a sales demo. Our methodological commitment is never to assert anything we cannot source to a public, citable page.
3. Vendor-by-vendor factual breakdown
3.1 Promotal MedConnect
Positioning: integrated professional telehealth kit, designed for primary care networks, community health centres, long-term care facilities, independent home-visit nurses and international deployments. Assembled in France in Ernée (Mayenne) by Groupe Eloi, a medical industrial group with 97 years of history.
Explicitly documented on the product page: 12-lead Bluetooth ECG Cardioline touchECG (with Schiller FT1, Schiller AT2+, Edan SE-1515 as documented alternatives), Riester Ri-Sonic digital stethoscope, multi-parameter bio-analyzer (glucose, cholesterol, lactate, triglycerides, uric acid), multi-lens medical camera (general practice, dermatoscope, otoscope), wireless vital signs monitor, spirometer, bladder scanner and dental camera in advanced configuration. Proprietary MedConnect software with AI medical scribe, Elara natural-language agent, and access to a tele-expertise network of 110,000+ professionals. Hosted on AWS with HDS certification, ISO 27001:2022 certified since June 2024, GDPR and HIPAA compliant.
Publicly disclosed price: starting from €3,000 ex-VAT for the base configuration, scaling with added instruments. Complete primary care network configurations and nursing home deployments sit above. See our MedConnect telehealth kit product page for the full datasheet and the complete 2026 buyer guide for procurement methodology.
Best-fit use case: organisations that want complete medical hardware + proprietary software + French and international regulatory compliance in a single purchase, without integrating multiple third-party tools.
3.2 Tessan
Positioning (source: info.tessan.io/mallette-de-teleconsultation, page consulted April 2026): French manufacturer targeting independent home-visit nurses in medically under-served areas, with the commercial message "Boost your revenue with medical teleconsultation".
What's explicitly documented on the product page: stethoscope, thermometer, otoscope, portable camera, Carte Vitale card reader. Proprietary Tessan software platform allowing the nurse to directly contact a Tessan physician for the teleconsultation.
What is not mentioned on the product page as of April 2026: no 12-lead ECG, no dermatoscope or bio-analyzer, no HDS hosting statement, no ISO 27001 claim, no public price, no AI scribe, no internal tele-expertise network. Some of these features may exist in their broader offer — but they are not surfaced on the landing page.
Best-fit use case: isolated independent home-visit nurses who want a simple, pre-configured solution with a commercial model where the remote physician is supplied by the vendor itself rather than by a partner network or the nurse's own referring clinic.
3.3 HEALPHI
Positioning (source: healphi.fr/mallette-telemedecine, page consulted April 2026): French manufacturer claiming over 300 units deployed, targeting primary care networks, nurses and medically under-served areas including French overseas territories and francophone Africa. The commercial message promotes a kit "priced lowest on the market", without disclosing a figure.
What's explicitly documented: stethoscope, otoscope, pulse oximeter, thermometer, blood pressure cuff (five instruments). Proprietary Healphi Web software with announced compatibility for Doctolib, Maiia, Qare. Hosted on "HDS servers" (the host itself is not publicly named on the page). Case described as "military-grade shock-resistant".
What is not mentioned on the product page as of April 2026: no 12-lead ECG, no dermatoscope, no bio-analyzer, no AI scribe, no ISO 27001, no public price despite the "cheapest" positioning.
Best-fit use case: organisations looking for a basic vital-instrument foundation at low cost, willing to delegate the teleconsultation software to a third-party platform such as Doctolib rather than relying on an integrated proprietary stack.
3.4 Hocoia — Hocobag
Important note: the Hocobag product page on hocoia.com was inaccessible (404 error) during our verification in April 2026. The site's "Our devices" page surfaces other Hocoia products (Médicobus, MammoBus) rather than the Hocobag kit. We cannot therefore factually document the current composition or feature set of the Hocobag from reliable public sources.
Recommendation: if Hocobag interests you, contact Hocoia directly to obtain a current product brochure and verify the commercial status of the reference before engaging in a purchase process.
3.5 PARSYS
Positioning (source: parsys.com and matelemedecine.fr, pages consulted April 2026): long-established French manufacturer (since 2010) specialising in the premium / emergency / field operations segment: emergency medical services, fire and rescue, maritime, mountain, aeronautical, medically-equipped long-term care. Two main form factors: the Station Ultra Portable (backpack) and the Station S2 (case).
What's explicitly documented: 12-lead Bluetooth ECG, blood pressure cuff, pulse oximeter, thermometer (optional), glucometer (optional). Proprietary Parsys Cloud + MedCapture + Real-Time Communication module. Hosting described as HDS-compliant (host not publicly named), two-factor authentication, CE-marked devices.
What is not mentioned on the product page as of April 2026: no specific dermatoscope, no AI scribe, no ISO 27001, no public price. The positioning is clearly premium, which explains the absence of entry pricing.
Best-fit use case: high operational-demand organisations (emergency medical services, fire and rescue, military, maritime) where ruggedness and reliability in extreme environments take priority over software flexibility or cost. Not a fit for a basic primary care network or a city-based independent nurse, where the form factor and likely price sit beyond the actual need.
3.6 Medeo Health (Kligo)
Positioning (source: medecins.medeo-health.com/infirmiers, page consulted April 2026): Medeo Health is an integrator, not strictly a kit manufacturer. The company offers its Kligo software and claims compatibility with 80+ connected medical devices. It describes two form factors: a "case" with six devices, and a "pouch" containing stethoscope + otoscope.
What's explicitly documented: compatible catalog including ECG, ultrasound, spirometer, blood pressure cuff, dermatoscope, glucometer and other connected instruments. Kligo software with announced integrations to the main French clinical information systems (Medistory, Weda, DrSanté and others).
What is not mentioned on the product page as of April 2026: no fixed, named "kit" SKU (the offer is an à-la-carte assembly rather than a single product), no explicit HDS hosting statement on the nurse-facing page we consulted, no AI scribe, no public price.
Best-fit use case: organisations that want to drive their own hardware assembly from a broad catalog, integrating into an existing clinical information system, without being locked into a fixed hardware bundle.
3.7 HOPI Medical — Wallet
Positioning (source: hopimedical.com, page consulted April 2026): French manufacturer offering a "Wallet" kit targeting home-visit nurses and medical practices. Commercial message: "Mobile solution · Highly compatible · Enhanced portability".
What's explicitly documented: fibre-optic otoscope, general-practice medical camera, dermatoscope, digital stethoscope, and ultrasonic doppler — a particularity rare on this segment. Announced compatibility with several third-party platforms: TeleMedica, eRosetta, Clinic alpha.
What is not mentioned on the product page as of April 2026: no 12-lead ECG, no proprietary software (the kit operates exclusively in compatibility mode with third-party platforms), no HDS hosting statement, no ISO 27001, no public price, no AI scribe.
Best-fit use case: a physician or practice that has already committed to a third-party teleconsultation platform and wishes to add an ultrasonic doppler to its remote examination capability — a function useful in obstetrics and vascular medicine that most other kits do not offer.
3.8 MediSpot ONE
Positioning (source: medispot.fr, page consulted April 2026): compact portable device (around 260g) that combines several functions in a single unit, paired with a MediSpot.Center SaaS platform. Target markets: general practice, pharmacies, sports medicine, home monitoring.
What's explicitly documented: thermometer, SpO₂, blood pressure cuff, HD camera (otoscope + dermatoscope lenses), cardio-pulmonary stethoscope. CE Class IIa marking, references to French data protection authority (CNIL), national health authority (HAS), GDPR. MediSpot.Center software platform with a public online shop (the price can therefore be consulted directly).
What is not mentioned on the product page as of April 2026: no 12-lead ECG, no bio-analyzer, no AI scribe, no explicit named HDS host, no ISO 27001. The form factor is that of a single portable all-in-one device rather than a true clinical kit with several distinct instruments.
Best-fit use case: practitioners looking for an ultra-portable, all-in-one tool for light general-practice or sports-medicine use — accepting a narrower range of clinical examinations compared to a true multi-instrument kit.
4. Which vendor fits which use case?
This section does not replace a quote or a sales demo. It synthesises, from the facts documented above, the scenarios where each vendor appears best-positioned — and those where they are less so.
- Independent home-visit nurse, simple clinical use case: HEALPHI, Tessan and HOPI Wallet explicitly position on this segment. MediSpot ONE can also fit if you don't need multiple distinct instruments. MedConnect is also suited, with the additional advantage of a 12-lead ECG and an AI scribe automatically generating the end-of-consultation note.
- Independent nurse handling complex cardiac cases: MedConnect is the only vendor to explicitly list a 12-lead Cardioline touchECG on its public product page, alongside PARSYS on the premium segment.
- Primary care network or community health center, multi-site coordinated deployment: MedConnect and HEALPHI are the two vendors who explicitly target this use case, with different philosophies (MedConnect on instrument breadth + complete software + AI; HEALPHI on entry cost + third-party platform compatibility). See our primary care network solution.
- Long-term care facility / nursing home: MedConnect, PARSYS and Medeo Kligo are the vendors that explicitly mention this use case. The choice depends on the key criterion: integrated software + AI scribe (MedConnect), emergency robustness (PARSYS), or à-la-carte assembly (Kligo). See our nursing home solution.
- Emergency medical services, fire and rescue, maritime, military: PARSYS is the explicit and historical positioning on this segment. MedConnect has experience on equivalent deployments (Hajj 2026 Saudi Arabia, New Caledonia, Ivory Coast) and is a credible alternative.
- Community pharmacy: Medeo Kligo and Tessan offer configurations specifically tailored to this use case. MediSpot ONE can also fit for light screening.
- International deployment (Africa, French overseas territories, Middle East): HEALPHI and MedConnect are the two vendors explicitly claiming deployments outside mainland France. MedConnect has to date documented deployments on four continents (Europe, Africa, Americas, Middle East). See our international solutions.
5. What distinguishes MedConnect in this comparison
We are both the author and a participant in this comparison — so we'll stay factual. Based on the data documented above, Promotal MedConnect is, in April 2026, the only French vendor to simultaneously deliver the following seven elements on its public product page:
- An explicitly named 12-lead Bluetooth ECG (Cardioline touchECG, with Schiller and Edan as documented alternatives).
- A named digital stethoscope (Riester Ri-Sonic), with eMurmur AI compatibility for cardiac-anomaly detection.
- A multi-parameter bio-analyzer with the exhaustive list of analyses (glucose, cholesterol, lactate, triglycerides, uric acid).
- An AI medical scribe with a conversational Elara agent integrated into the platform, for automatic SOAP note generation in natural language.
- A tele-expertise network of 110,000+ professionals accessible directly from the platform, with no third-party software in the loop.
- An explicitly named AWS host with HDS certification, plus a verifiable ISO 27001:2022 certification (obtained June 2024).
- A publicly disclosed starting price (from €3,000 ex-VAT), unusual in the French telehealth kit market.
This combination is also assembled in France in Ernée (Mayenne) by Groupe Eloi, a centenarian medical industrial group, with verified deployments on four continents (mainland France, New Caledonia, Ivory Coast, United States, Saudi Arabia — Hajj 2026 deployment in progress with 60 kits). More than 50,000 examinations have been performed on the MedConnect platform, a clinical volume that feeds the embedded AI algorithms.
This does not mean MedConnect is the best choice for every use case — that would contradict the methodology of this article. If you're an isolated nurse with a very tight budget and simple clinical cases, HEALPHI or Tessan may be enough. If you're an emergency medical service, PARSYS has specific tenure on the emergency segment. If you're already engaged with a third-party platform and just want to complement your instruments, Medeo Kligo or HOPI Wallet offer à-la-carte assemblies.
But if you are looking for a complete, integrated, compliant, pricing-transparent solution ready for multi-site deployment across multiple user profiles — then, as of April 2026, it is the solution we document most thoroughly in this comparison.
6. Next steps
Before deciding, three concrete actions:
- Read our complete 2026 telehealth kit buyer guide — it contains the 12-criterion checklist that lets you evaluate any vendor beyond this matrix.
- Ask every shortlisted vendor the same structured questions: exact list of included instruments with brand and model, name of the HDS-certified host, date of ISO 27001 certification, all-inclusive 3-year total cost (hardware + software + maintenance). A vendor evasive on any of these points is a signal.
- Download the Promotal MedConnect catalog as a PDF if you want to compare our full datasheet against competitors — no sales call involved.
7. Frequently asked questions
Why doesn't this comparison include Doctolib, Maiia or Qare?
Because Doctolib, Maiia and Qare are not telehealth kit manufacturers. They are video-consultation and scheduling platforms. They do not integrate connected medical hardware (ECG, stethoscope, bio-analyzer), so they do not address the same clinical need as a telehealth kit. They are complementary, not competing.
Why don't US giants (Teladoc, Amwell, Caregility) appear?
These players are positioned on different segments: Teladoc and Amwell on B2C/B2B SaaS teleconsultation, Caregility on in-room hospital telehealth. None of them actively commercialise a mobile clinical kit in the French market as of April 2026. We preferred to restrict this comparison to vendors actually present on the French telehealth kit segment.
Is the matrix biased in favour of MedConnect?
We are MedConnect — so the question is legitimate. Our methodological commitment is the following: every "✓ Yes" for MedConnect can be verified on our telehealth kit product page, our software platform page and our Vanta trust center. Every "— Not mentioned" for a competitor means only that the information was not on their public product page in April 2026 — not that it does not exist. If a competitor believes we have missed factual information, we correct on demand at [email protected].
How much does a kit cost in each tier?
None of the compared vendors publishes a complete price schedule. The only public information we can factually confirm is MedConnect's (from €3,000 ex-VAT for the base configuration) and MediSpot ONE's (online shop, public price). For the others, a quote is required. Our 2026 buyer guide publishes price ranges observed in the market (€2,500 to €30,000+ ex-VAT plus subscription options) to give you an order of magnitude.
Will this comparison be updated?
Yes. We revise this type of content at least twice a year and we accept corrections or additions from vendors we missed. If your company is cited and you want to correct an entry or complete your profile, write to us at [email protected] — we will document the update with the new consultation date.
8. Methodology disclaimer
This comparison is an editorial / journalistic work intended for information purposes, written by the Promotal MedConnect team in April 2026. Information about our competitors comes exclusively from their public product pages, consulted between April 1st and April 15th, 2026. "Not mentioned" signals only that the information was not visible on the public page at the time of consultation — it may exist in a PDF brochure, a custom quote, or a private sales presentation. Brand and product names remain the property of their respective owners. No external clickable link points to competitor websites, in order to respect their commercial independence and avoid any ambiguous association. If you are a cited vendor and you want to correct, complete or withdraw your profile, please write to [email protected] — we will document the update with the date.
Ready to discover MedConnect?
Request a personalized demo and see how the platform adapts to your practice.
Request a demo